March 1, 2020
By Debbie Jones, BSc, FCOptom, FAAO
 Myopic patients must be managed and monitored closely. Myopia can no longer be considered as a benign refractive error, and it cannot be managed by traditional optical means. With the growing prevalence of myopia1, and the increased risk of ocular pathology associated with high myopia2,3, eye care professionals (ECPs) need to assess their patients for risk factors that may lead to its onset.
Myopic patients must be managed and monitored closely. Myopia can no longer be considered as a benign refractive error, and it cannot be managed by traditional optical means. With the growing prevalence of myopia1, and the increased risk of ocular pathology associated with high myopia2,3, eye care professionals (ECPs) need to assess their patients for risk factors that may lead to its onset.
Axial length
Axial length (AL) is the combination of anterior chamber depth, lens thickness and vitreous chamber depth, and it is the most significant contributor to refractive error.4 Myopia results from an increase in AL outside of the normal rate expected for age. Typically, AL increases from around 16.8 mm in infancy to 23.6 mm in adulthood, with that increase being offset by changes in the refractive state of the other ocular components, primarily the crystalline lens and cornea.5 A change in AL of 1 mm with no other ocular compensation would, for example, result in a myopic shift of around 2.00D to 2.50D. In an adult myopic population, a bimodal distribution has been demonstrated, with the first peak at an AL of 24 mm for low myopia (≥ – 6.00D) and a second peak at an AL of 30 mm for high myopia (< -6.00D).6 Further, it is well accepted that the onset of myopia prior to the age of 10 results in a higher risk of high myopia in adulthood.7,8
Change in AL is influenced by age, race and baseline refractive error. Younger children change at a more rapid rate, which slows with age.4,9,10 A typical annual expected change in AL for a young emmetropic child under the age of eight is around 0.20 mm per year. Myopic children demonstrate more rapid AL growth than both emmetropic and hyperopic children (average of 0.34 mm per year), with a correlation between axial length and refractive error. This slows to around 0.10 mm to 0.15 mm per year between the ages of 9 and 11, which continues to slow through puberty.9,11 Asian children, in general, demonstrate a faster rate of increase in AL than Caucasian counterparts.11,12
Recently published work has provided some insight into normal growth rates, enabling the risk of myopia to be assessed9, with normative values being published for a cohort of European children. The collected data were reported as percentiles, a term that is well understood for other growth factors such as height and weight. Figure 1 illustrates the results of the study and demonstrates that children below the 50th percentile for AL increase are significantly less likely to become myopic (and almost certainly not be highly myopic), compared to those above the 50th percentile.
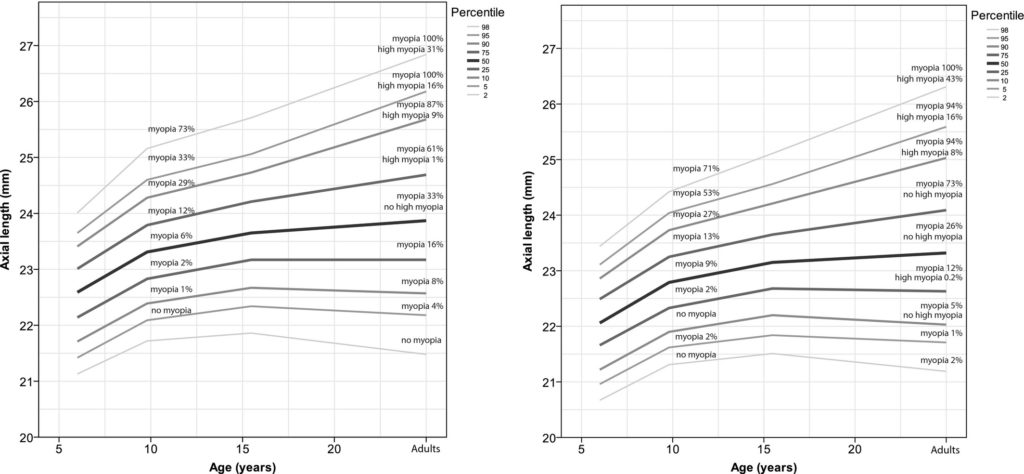
Figure 1 – Axial length vs. age (males left, females right), with myopia risk assessment9
A comparable study in Asia found similar results, although the AL measured at each percentile were higher, indicating the higher prevalence of myopia in that population. For example, in the European population, 15-year-old females had AL values of 23.15 mm for the 50th percentile, whereas the Asian population had a mean AL of 24.37 mm for an age-matched sample.10
The availability of percentile data on large population samples should help ECPs match patient age, refractive error and AL to a predicted outcome. This illustrates the importance of being able to measure AL in myopia management.
Measuring Axial Length
AL measurement has not traditionally been considered part of mainstream optometry. The same could have been said for visual field assessment four decades ago and for using an OCT a decade ago. Just as primary care practice has moved forward with these two examples, it should be the aim of the profession to do the same for AL measurement in myopia management.
There are two main options for measuring AL: ultrasound biometry and optical biometry. A-scan ultrasound biometry is a more challenging technique to use on children due to the contact nature of the instrument. Within these two methodologies, there are many options available for optical biometry, covering a range of budget options. Most instruments are easy to use on children and are non-invasive.
Whether AL measurement is a clinical necessity is something that has been and continues to be widely debated. The IMI Clinical Management Guidelines report recommends that “If available, axial length measurements should be taken at least every six months” with an acknowledgment that AL measurement is an expected outcome measure of a clinical trial.13 That begs the question, if AL is an essential outcome measure for a clinical trial, then why shouldn’t it be an essential outcome measure for clinical practice in myopia management?
Value of Axial Length in Monitoring Myopia Progression
Having committed to a myopia control strategy for a patient, it is necessary to monitor that patient to determine whether the chosen treatment is slowing the myopia progression. Progression is not just the change in refractive error over time; it is also the change in AL. Measuring and monitoring only one of the components could be considered to be doing only half a job.
Published literature has demonstrated that it is easy to be fooled into thinking that your chosen myopia treatment is having the desired effect when the refractive error is being assessed. The atropine-based ATOM2 study showed very positive, and similar, refractive error outcomes for all concentrations evaluated, leading to the conclusion that the lower concentration of 0.01% atropine, with its less severe side effects, was the best starting option for use in practice. However, further analysis has led to an understanding that the change in refractive error and AL were not well correlated.14,15 This has resulted in updated recommendations of starting with higher concentrations of atropine, such as 0.02% or higher.16 If orthokeratology is the chosen treatment strategy, the measurement of AL is the only method to monitor the effect of the treatment, given that the measured refractive error is effectively eliminated by the treatment.17,18
Monitoring the effect of any myopia management intervention is crucial. One way of achieving this is to collect accurate data over time, with the collated data then used to confirm whether a treatment strategy is working or not. This can help with the initial phase of getting reluctant parents on board with the need to implement treatment, along with informing discussions over follow-up visits once an intervention has been prescribed. When AL measurements can support this communication, the evidence shared with parents becomes more compelling and credible.
Parent Communication
When parents are reluctant to enter the realm of myopia management, often stating that simple correction has been perfectly suitable for themselves, ECPs need to find a way to convince them that there is good evidence to be proactive and intervene early. Axial elongation, and subsequent increase in myopia, results in an increased risk of ocular complications associated with myopia.19 Being able to point to their child’s AL measurement and show the likely trajectory and the associated risks provides compelling evidence that intervention is appropriate.
Left unmanaged, high myopia can impact occupational choices in later life. For example, emergency services personnel and military recruits are typically required to have a reasonable level of uncorrected acuity, limiting career options for even moderate myopes. It has been shown that patients with high degrees of myopia experience a poorer quality of life than those with low to moderate myopia20. Refractive surgery can also be affected, with outcomes generally better in patients with lower refractive errors.21 These factors may help to convince parents of the need to enter into a myopia management program to help change the future of their child’s myopia progression and reduce the risk of future complications that could lead to visual impairment.
The debate over the necessity of monitoring AL in routine practice will continue, and it is generally accepted that starting any form of myopia management, even in the absence of AL measures, is better than taking no action at all. However, the evidence already exists to demonstrate the importance of obtaining an AL measurement regularly and relating it to the patient’s refractive error. AL measurement should be considered the gold standard of myopia management, and investing in the equipment required to measure AL is ultimately an investment in the future of your patients.
References
- Holden BA, Fricke TR, Wilson DA, Jong M, Naidoo KS, Sankaridurg P, et al. Global Prevalence of Myopia and High Myopia and Temporal Trends from 2000 through 2050. Ophthalmology. 2016;123(5):1036-42.
- Flitcroft DI. The complex interactions of retinal, optical and environmental factors in myopia aetiology. Prog Retin Eye Res. 2012;31(6):622-60.
- Jones D, Luensmann D. The prevalence and impact of high myopia. Eye Contact Lens. 2012;38(3):188-96.
- Mutti DO, Hayes JR, Mitchell GL, Jones LA, Moeschberger ML, Cotter SA, et al. Refractive error, axial length, and relative peripheral refractive error before and after the onset of myopia. Invest Ophthalmol Vis Sci. 2007;48(6):2510-9.
- Gordon RA, Donzis PB. Refractive development of the human eye. Arch Ophthalmol. 1985;103(6):785-9.
- Tron E. The optical elements of the refractive power of the eye. In: F. Ridley, Sorsby A, editors. Modern Trends of Ophthalmology. London: Butterworth; 1940.
- Parssinen O, Kauppinen M. What is the influence of parents’ myopia on their children’s myopic progression? A 22-year follow-up study. Acta Ophthalmol. 2016;94(6):579-85.
- Tideman JWL, Polling JR, Hofman A, Jaddoe VW, Mackenbach JP, Klaver CC. Environmental factors explain socioeconomic prevalence differences in myopia in 6-year-old children. Br J Ophthalmol. 2018;102(2):243-7.
- Tideman JWL, Polling JR, Vingerling JR, Jaddoe VWV, Williams C, Guggenheim JA, et al. Axial length growth and the risk of developing myopia in European children. Acta Ophthalmol. 2018;96(3):301-9.
- Sanz Diez P, Yang LH, Lu MX, Wahl S, Ohlendorf A. Growth curves of myopia-related parameters to clinically monitor the refractive development in Chinese schoolchildren. Graefes Arch Clin Exp Ophthalmol. 2019;257(5):1045-53.
- Jones LA, Mitchell GL, Mutti DO, Hayes JR, Moeschberger ML, Zadnik K. Comparison of ocular component growth curves among refractive error groups in children. Invest Ophthalmol Vis Sci. 2005;46(7):2317-27.
- Wong HB, Machin D, Tan SB, Wong TY, Saw SM. Ocular component growth curves among Singaporean children with different refractive error status. Invest Ophthalmol Vis Sci. 2010;51(3):1341-7.
- Gifford KL, Richdale K, Kang P, Aller TA, Lam CS, Liu YM, et al. IMI – Clinical Management Guidelines Report. Invest Ophthalmol Vis Sci. 2019;60(3): M184-M203.
- Bullimore MA, Berntsen DA. Low-Dose Atropine for Myopia Control: Considering All the Data. JAMA Ophthalmol. 2018;136(3):303.
- Khanal S, Phillips JR. Which low-dose atropine for myopia control? Clin Exp Optom. 2019.
- Yam JC, Jiang Y, Tang SM, Law AKP, Chan JJ, Wong E, et al. Low-Concentration Atropine for Myopia Progression (LAMP) Study: A Randomized, Double-Blinded, Placebo-Controlled Trial of 0.05%, 0.025%, and 0.01% Atropine Eye Drops in Myopia Control. Ophthalmology. 2019;126(1):113-24.
- Na M, Yoo A. The effect of orthokeratology on axial length elongation in children with myopia: Contralateral comparison study. Jpn J Ophthalmol. 2018;62(3):327-34.
- Zhu MJ, Feng HY, He XG, Zou HD, Zhu JF. The control effect of orthokeratology on axial length elongation in Chinese children with myopia. BMC Ophthalmol. 2014;14:141.
- Xiao O, Guo X, Wang D, Jong M, Lee PY, Chen L, et al. Distribution and Severity of Myopic Maculopathy Among Highly Myopic Eyes. Invest Ophthalmol Vis Sci. 2018;59(12):4880-5.
- Rose K, Harper R, Tromans C, Waterman C, Goldberg D, Haggerty C, et al. Quality of life in myopia. Br J Ophthalmol. 2000;84(9):1031-4.
- Bailey MD, Olson MD, Bullimore MA, Jones L, Maloney RK. The effect of LASIK on best-corrected high-and low-contrast visual acuity. Optom Vis Sci. 2004;81(5):362-8.

Debbie Jones, BSc, FCOptom, FAAO, is a clinical professor at the School of Optometry and Vision Science and a clinical scientist at the Centre for Ocular Research & Education (CORE) at the University of Waterloo. Her main area of clinical focus is in pediatric optometry, and her primary area of research activity is in the field of myopia management.