April 15, 2019
By Maria Liu, OD, PhD, FAAO
Associate Professor of Clinical Optometry
UC Berkeley, School of Optometry
Myopia is becoming a worldwide epidemic and imposes a tremendous public health burden.1-4 On top of the drastic increase in the overall prevalence of myopia, it is more concerning that the age of onset has become much younger, resulting in a faster progression, older age of myopia stabilization, higher degree of myopia, and significantly increased risks of complications that may lead to irreversible vision loss.5-7 While there have been significant advancements in the understanding of the etiology of the condition, as well as the efficacies of some evidence-based myopia control options, much of the clinical research efforts have been focused on children with a lower degree of myopia. Unlike what had been previously observed in earlier cohorts that myopia tends to stop progressing around the age of 15,8 it is not uncommon to see patients with continuous myopic progression into their 30s, especially in Asian ethnicity.
When does myopia stop progressing?
One of the most commonly asked questions by the parents during the initial myopia management consultation is how long their children will need to continue with their myopia treatments such as overnight orthokeratology, daytime multifocal contact lens, or nighttime low dose atropine drops. In other words, when does myopia stop progressing? While the age of onset remains as the primary predictor of the age of myopia stabilization and the level of myopia at the endpoint of progression,9 other factors such as the continuously changing lifestyle and visual demand cannot be ignored. For example, it is more common to see myopic progression among graduate or professional students of the same age compared to those who have less near workload and more outdoor exposure. Additionally, while myopic progression and axial ocular growth tend to slow down with age, it is possible to have a recurrent phase of progression even in the adulthood.10-15 At UC Berkeley School of Optometry Myopia Control Clinic, we have encountered more than a dozen of patients who had some level of progression after years of stable myopia, most commonly triggered by a drastic change of profession or lifestyle. As a result, it is essential to keep in mind that myopia management is a long-term and dynamic effort and the treatment strategies need to be frequently re-evaluated and adjusted if necessary, to match the patients’ constant change of visual demands and risk exposure.
Does adult progressive myopia share the same etiology and responds equally to the myopia control treatments as juvenile myopia?
It is commonly accepted that myopia is a combined result of genetic predisposition and environmental influence. However, the complicated interactions between the two remain to be elucidated.6,16 For myopia that continues to worsen throughout adulthood, it tends to be more prone to genetics, present in higher severity, longer axial length, and more degenerative signs in the posterior ocular structures.17-19 With continuous axial elongation and the excessive stretch of the posterior sclera, and the resulting compromised support to the overlying structures such as choroid, Bruch’s membrane, RPE, and ultimately the outer retina, the risks of degenerative and traction myopic retinopathy increase exponentially.16-19 While there is some evidence supporting myopia controlling efficacies of low dose atropine and overnight OrthoK in high myopia;20,21 it is extremely challenging to maintain the functional and anatomical stability of posterior ocular structure once the scleral stretching and thinning reach its decompensating limit, regardless of the age of the patient and the intensity of near work. Furthermore, the prognosis for degenerative myopia reduces significantly in the presence of posterior staphyloma.22 Consequently, early interventions that target slowing down myopic progression and the excessive scleral stretching play crucial role in minimizing the risks of subsequent ocular complications.
Clinical considerations for selecting treatments for myopia retardation in adult patients
Managing adult progressive myopes impose some unique challenges mostly due to their higher visual demands. Despite the excellent tolerance of both overnight OrthoK and daytime multifocal contact lenses in pediatric patients as the mainframe myopia control treatments, the unwanted glare and halo associated with both options may reduce their acceptance in adult myopes significantly.23 Similarly, with low dose atropine, which only has mild impacts on the light sensitivity and near vision in children, can be quite intolerable in adult population due to their reduced accommodative amplitude hence less buffer to near blur.24,25 As a result, adult patients must receive intensive education that emphasizes the importance of myopia control and axial growth inhibition as well as the expected side effects of the options. This education is necessary before the initiation of treatment to allow maximum understanding of treatment goals and realistic expectations of the visual performance.
For adult patients presenting with continuous axial elongation and signs of myopic retinopathy in dilated exams, fundus photo documentation and baseline imaging by high frequency B-scan ultrasonography and OCT are crucial in detecting subtle pathological changes and making a timely referral. OCT provides high-resolution images of posterior ocular structures in both the coronal and sagittal planes, making it the test of choice for monitoring myopic maculopathy. However, its field of view is somewhat limited for the detection of posterior abnormality in the central 16mm region, and the cost of the instrument could be inhibiting for some practices. Additionally, in myopic eyes with deep staphyloma, cross-sectional OCT images are often distorted, making it more difficult to interpret.26 Consequently, high frequency B-scan ultrasonography serves as a portable and affordable complementary test to fundus photo and OCT in evaluating myopic complications both centrally and peripherally (Figure 1.).
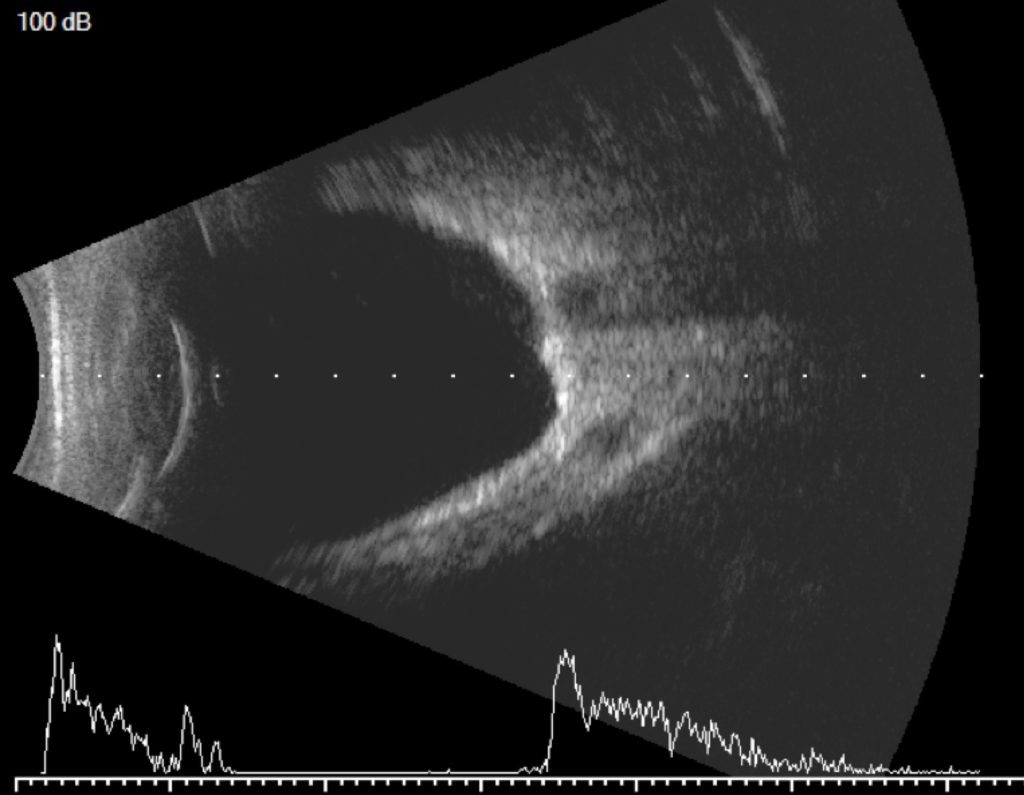
Figure 1. High-frequency B-scan ultrasonography showing the posterior staphyloma of a highly myopic eye.
In addition to the non-invasive optical and pharmaceutical options for myopia management, posterior-pole scleral buckling has shown promising results in controlling the persistent axial elongation in patients with advanced degenerative myopia such as macular schisis.17,18 If appropriately positioned, the buckle provides strong support to the stretched sclera and force it to come forward and proximal to the overlying choroid, and the posterior retina, hence closing the schisis cavity. IOP lowering medications such as beta-blockers have also been used empirically in the attempt to reduce the interocular drive of posterior scleral stretching. However, the results were equivocal in the more rigorous clinical trial testing.27
Summary
Not only the overall prevalence of myopia is increasing rapidly worldwide, but it is also alarming that the age of onset of myopia has become younger, and the age of myopia stabilization has become older, resulting in a much higher risk of fast progression and the development of high myopia. Although myopia progression tends to slow down with age, it is not uncommon to observe continuous progression and axial elongation into adulthood. Overnight OrthoK, as well as topical atropine, have shown controlling effects in high myopia however extensive patient education is necessary to ensure a better understanding of treatment priority and realistic expectations of visual performance provided by the myopia control options. Finally, myopia management is a long-term and continuous effort. In addition to early interventions that focus on slowing the myopic progression and axial elongation in younger children, comprehensive fundus exams, detailed evaluation of posterior ocular health, as well as timely referral to a retina specialist are critical components in managing adult high myopia.
References
- Holden BA, Fricke TR, Wilson DA, et al. Global prevalence of myopia and high myopia and temporal trends from 2000 through 2050. Ophthalmology. 2016; 123:1036–1042.
- Williams KM, Bertelsen G, Cumberland P et al.; European Eye Epidemiology (E(3)) Consortium. Increasing prevalence of myopia in Europe and the impact of education. Ophthalmology 2015; 122: 1489–1497.
- Lam CS, Lam CH, Cheng SC & Chan LY. Prevalence of myopia among Hong Kong Chinese schoolchildren: changes over two decades. Ophthalmic Physiol Opt 2012; 32: 17–24.
- Pan CW, Dirani M, Cheng CY, Wong TY & Saw SM. The age‐specific prevalence of myopia in Asia: a meta‐ Optom Vis Sci 2015; 92: 258–266.
- Grossniklaus HE, Green WR. Pathologic findings in pathologic myopia. Retina. 1992; 12:127–133.
- Morgan IG, Ohno-Matsui K, Saw SM. Myopia. Lancet 2012; 379: 1739–1748
- Walline JJ, Gaume A, Jones LA, et al. Benefits of contact lens wear for children and teens. Eye Contact Lens. 2007; 33:317–321.
- The COMET Group. Myopia Stabilization and Associated Factors Among Participants in the Correction of Myopia Evaluation Trial (COMET). Invest Ophthalmol Vis Sci. 2013 Dec; 54(13): 7871–7884.
- Chua, SYL, Sabanayagam, C, Cheung, Y‐B, Chia, A, Valenzuela, RK, Tan, D, Wong, TY, Cheng, C‐Y, Saw, S‐M. Age of onset of myopia predicts risk of high myopia in later childhood in myopic Singapore children. Ophthalmic Physiol Opt2016; 36: 388– 394.
- Parssinen O, Lyyra AL. Myopia and myopic progression among schoolchildren: A three-year follow-up study. Invest Ophthalmol Vis Sci 1993; 34:2794–2802.
- Goss DA. Near work and myopia. Lancet 2000; 356:1456–1457.
- Hepsen IF, Evereklioglu C, Bayramlar H. The effect of reading and near-work on the development of myopia in emmetropic boys: A prospective, controlled, three-year follow-up study. Vision Res 2001; 41:2511–2520.
- Saw SM, Chua WH, Hong CY, et al. Near work in early-onset myopia. Invest Ophthalmol Vis Sci 2002; 43:332–339.
- Saw SM, Wu HM, Seet B, et al. Academic achievement, close up work parameters, and myopia in Singapore military conscripts. Br J Ophthalmol 2001; 857:855–860.
- Li SM, Li SY, Kang MT, et al. Near work related parameters and myopia in Chinese children: The Anyang Childhood Eye study. PLoS One 2015;10: e0134514.
- Flitcroft DI. The complex interactions of retinal, optical and environmental factors in myopia aetiology. Prog Retin Eye Res 2012; 31:622–660.
- Ward B. Tarutta EP. Mayer MJ. The efficacy and safety of posterior pole buckles in the control of progressive high myopia.Eye (Lond).2009 Dec;23(12):2169-74.
- Ward B. Degenerative Myopia: Myopic Macular Schisis and the Posterior Pole Buckle. Retina. 2013 Jan;33(1):224-31.
- Saw SM, Gazzard G, Shih-Yen EC, Chua WH. Myopia and associated pathological complications. Ophthal Physiol Opt. 2005; 25:381–391.
- Charm J. Cho P. High Myopia–Partial Reduction Ortho-k: A 2-Year Randomized Study. Optometry and Vision Science. 2013;90(6):530–539.
- Chou AC, Shih YF, Ho TC, Lin LL. The effectiveness of 0.5% atropine in controlling high myopia in children. J Ocul Pharmacol Ther. 1997;13(1):61–7.
- Steidl SM, Pruett RC. Macular complications associated with posterior staphyloma. Am J Ophthalmol 1997; 123:181– 187.
- Kang P, McAlinden C, Wildsoet CF. Effects of multifocal soft contact lenses used to slow myopia progression on quality of vision in young adults. Acta Ophthalmol 2017;95: e43-e53.
- Kaymak H, Fricke A, Mauritz Y, et al. Short-term effects of low-concentration atropine eye drops on pupil size and accommodation in young adult subjects. Albrecht von Graefe’s Archiv für Ophthalmologie. 2018;256(12):1-7
- N Kona, SE Kochik, Y Liu.8-Hour Survey of 0.01% Atropine-Induced Changes in Pupil Size and Accommodative Function. (Poster presentation) AAO 2018
- Schuman J. Optical Coherence Tomography in High Myopia. JAMA Ophthalmol. 2016 Sep 1; 134(9): 1040.
- Jensen H. Myopia progression in young school children. A prospective study of myopia progression and the effect of a trial with bifocal lenses and β blocker eye drops. Acta Ophthalmol. Suppl. 1991; 200:1–79.