May 3, 2021
By Pauline Kang, B.Optom, PhD
School of Optometry and Vision Science, University of New South Wales
Pharmaceutical myopia control interventions such as low concentration atropine are another option for patients deemed unsuitable for toric OrthoK or multifocal soft contact lenses, as refractive error doesn’t impact patient suitability for pharmaceutical treatments.

Myopia control options for children with high refractive astigmatism are growing with the development of new contact lens designs for correcting higher degrees of refractive astigmatism. However, clinical trials evaluating the efficacy of these treatments are limited, and it is often assumed that myopia control effects with optical interventions such as orthokeratology (OrthoK) will be similar between progressive myopic children with and without significant refractive astigmatism.
Toric Orthokeratology
OrthoK involves overnight wear of rigid gas-permeable contact lenses that temporarily change corneal topography to correct ametropia. In myopic OrthoK, there is central corneal flattening and mid-peripheral corneal steepening.1,2 Clinical trials have demonstrated that OrthoK not only corrects myopia but also slows it’s progression in children.3-7 Recent meta-analyses have reported an overall 41 to 45 percent or 0.25 to 0.27 mm reduction in axial elongation in children fitted with OrthoK compared to single vision correction over a two-year treatment period.8-10 The inclusion criteria of OrthoK clinical trials have generally required children to have up to 4.00D of myopia and less than 1.50D of refractive astigmatism.3-7 The refractive astigmatism criteria ensures participants have minimal corneal toricity when fitted with spherical back optic zone OrthoK lenses in the clinical trials, as there is a significant correlation between refractive astigmatism and corneal toricity.11,12
Spherical OrthoK lens designs incorporate four or five zones of curves that are rotationally symmetrical. When applied to a highly toric cornea (greater than 1.50D of corneal toricity), lens and subsequent treatment zone decentration occur, resulting in poor visual outcomes.11 Toric OrthoK lens designs were developed to reduce refractive astigmatism and improve lens and treatment zone centration on highly toric corneas. Toric OrthoK lenses incorporate either a spherical or toric central base curve with adjacent toric reverse, alignment, or peripheral curve zones. This results in different lens sagittal heights along orthogonal meridians to better align the OrthoK lens with the patient’s toric cornea.2 It is estimated that 1.00D of corneal toricity is equivalent to a 25µm difference in corneal elevation along the 8mm corneal chord.12
As previous clinical trials involved children with only mild degrees of astigmatism, the Toric Orthokeratology – Slowing Eyeball Elongation (TO-SEE) study was conducted to evaluate myopia control efficacy with toric OrthoK. This prospective study involved 6- to 12-year-old children with myopia between -0.50D to -5.00D and with-the-rule refractive astigmatism between 1.25D to 3.50D (axis 180 ± 20) who self nominated to be corrected with either OrthoK or single vision spectacles over two years. Children in the OrthoK group were fitted with toric OrthoK lenses (Menicon Z Night Toric design). Thirty-five out of the initial 43 participants recruited in the OrthoK group and 23 of the initial 37 subjects recruited in the control single vision group completed the two-year clinical trial. A mean increase in axial length of 0.31 ± 0.27 mm and 0.64 ± 0.31 mm in the OrthoK and control groups, respectively, indicated just over 50 percent reduction in myopia progression with toric OrthoK treatment.
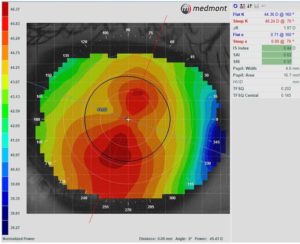
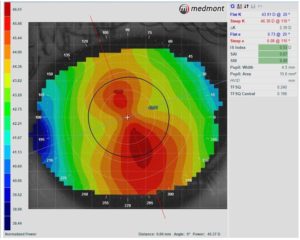 Two retrospective studies have compared the effects of spherical and toric OrthoK lens designs on myopia progression with mixed results. Zhang et al. found that children with moderate corneal astigmatism (1.50D to 3.50D with-the-rule) fitted with a toric OrthoK lens had significantly less myopia progression than children fitted with spherical OrthoK lens (toric: 0.04 ± 0.13 mm versus spherical: 0.09 ± 0.13 mm) in one year.13 In contrast, Jiang et al. found no difference in myopia control effects between spherical and toric lens designs (toric: 0.13 ± 0.18 versus spherical: 0.11 ± 0.20).14
Two retrospective studies have compared the effects of spherical and toric OrthoK lens designs on myopia progression with mixed results. Zhang et al. found that children with moderate corneal astigmatism (1.50D to 3.50D with-the-rule) fitted with a toric OrthoK lens had significantly less myopia progression than children fitted with spherical OrthoK lens (toric: 0.04 ± 0.13 mm versus spherical: 0.09 ± 0.13 mm) in one year.13 In contrast, Jiang et al. found no difference in myopia control effects between spherical and toric lens designs (toric: 0.13 ± 0.18 versus spherical: 0.11 ± 0.20).14
What to Prescribe a Myopia Patient with Moderate to High Astigmatism
Currently, we are unable to predict which myopia control treatment will be best suited for individual patients. Results from OrthoK clinical trials are mean responses, and there is variability in myopia control achieved between participants. In clinical practice, myopia control treatments should be selected based on individual patient suitability and preferences, and myopia progression should be monitored over regular periods to assess for treatment efficacy.2,15 The International Myopia Institute developed clinical guidelines for various myopia control treatments,15 and in March 2021, the British Contact Lens Association published the Contact Lens Evidence-based Academic Report (CLEAR) – Orthokeratology, a detailed review on OrthoK including its use in myopia management.2
Toric OrthoK lenses can be prescribed to myopic patients with high astigmatism; if the refractive astigmatism is mostly corneal in nature and depending on the limit of lens toricity for the selected OrthoK lens design, then toric OrthoK lenses may be a suitable option for the patient.
Toric multifocal soft contact lenses with center distance designs are another potential treatment option. However, no studies to date have investigated the impact of toric multifocal soft contact lenses on myopia progression. Clinical trials of spherical multifocal soft contact lenses (aspherical/progressive, dual focus/concentric, or extended depth of focus designs) have reported promising myopia control results,16-19 often inferred to patients with high refractive astigmatism fitted with toric multifocal lenses. These studies have included spherical lens designs only and fitted on children with <1.00D of refractive astigmatism.
Pharmaceutical myopia management interventions such as low concentration atropine are another option for patients deemed unsuitable for toric OrthoK or multifocal soft contact lenses, as refractive error doesn’t impact patient suitability for pharmaceutical treatments.
Future Work
Myopia management studies are limited to children with low to moderate myopia and minimal astigmatism. With more complex customized OrthoK lens designs available to fit astigmatic myopic patients, further studies confirming the myopia control effects of toric OrthoK lenses in myopic children with moderate to high degrees of refractive astigmatism are required.
A previous study investigated partial correction of high myopia with OrthoK and residual refractive errors corrected with daytime spectacles.20 This may be a potential option for high astigmatic patients in OrthoK with residual astigmatism. However, clinical trials are required to confirm myopia control efficacy with partial astigmatic correction.

Pauline Kang, B.Optom, PhD, is a Senior Lecturer at the School of Optometry and Vision Science, University of New South Wales, Sydney, Australia. Dr. Kang’s research focuses on optical strategies for myopia control.
References
- Swarbrick HA. Orthokeratology review and update. Clin Exp Optom 2006;89:124-143.
- Vincent SJ et al. CLEAR – Orthokeratology. Cont Lens Anterior Eye 2021;44:240-269.
- Cho P, Cheung SW. Retardation of Myopia in Orthokeratology (ROMIO) study: A 2-year randomized clinical trial. Invest Ophthalmol Vis Sci 2012;53:7077-7085.
- Cho P et al.. The longitudinal orthokeratology research in children (LORIC) in Hong Kong: a pilot study on refractive changes and myopic control. Curr Eye Res 2005;30:71-80.
- Walline JJ et al. Corneal reshaping and myopia progression. Br J Ophthalmol 2009;93:1181-1185.
- Santodomingo-Rubido J et al. Myopia control with orthokeratology contact lenses in Spain: refractive and biometric changes. Invest Ophthalmol Vis Sci 2012;53:5060-5065.
- Kakita T et al. Influence of overnight orthokeratology on axial elongation in childhood myopia. Invest Ophthalmol Vis Sci 2011;52:2170-2174.
- Si JK et al. Orthokeratology for myopia control: a meta-analysis. Optom Vis Sci 2015;92:252-257.
- Sun Y et al. Orthokeratology to control myopia progression: a meta-analysis. PLoS One 2015;10:e0124535.
- Wen D et al. Efficacy and Acceptability of orthokeratology for slowing myopic progression in children: a systematic review and meta-analysis. J Ophthalmol 2015;2015:360806.
- Cheung SW, Cho P, Chan B. Astigmatic changes in orthokeratology. Optom Vis Sci 2009;86:1352-1358.
- Tomiyama ES et al. Corneal elevation, power, and astigmatism to assess toric orthokeratology lenses in moderate-to-high astigmats. Eye Contact Lens 2021;47:86-90.
- Zhang Y, Chen YG. Comparison of myopia control between toric and spherical periphery design orthokeratology in myopic children with moderate-to-high corneal astigmatism. Int J Ophthalmol 2018;11:650-655.
- Jiang J et al. Comparison of toric and spherical orthokeratology lenses in patients with astigmatism. J Ophthalmol 2019;2019:4275269.
- Gifford KL et al. IMI – Clinical Management Guidelines Report. Invest Ophthalmol Vis Sci 2019;60:M184-M203.
- Li SM et al. Studies using concentric ring bifocal and peripheral add multifocal contact lenses to slow myopia progression in school-aged children: a meta-analysis. Ophthalmic Physiol Opt 2017;37:51-59.
- Walline JJ et al. Effect of high add power, medium add power, or single-vision contact lenses on myopia progression in children: the BLINK randomized clinical trial. JAMA 2020;324:571-580.
- Chamberlain P et al. A 3-year randomized clinical trial of MiSight lenses for myopia control. Optom Vis Sci 2019;96:556-567.
- Sankaridurg P et al. Myopia control with novel central and peripheral plus contact lenses and extended depth of focus contact lenses: 2 year results from a randomized clinical trial. Ophthalmic Physiol Opt 2019;39:294-307.
- Charm J, Cho P. High myopia-partial reduction ortho-k: a 2-year randomized study. Optom Vis Sci 2013;90:530-539.













