March 13, 2019
By Erin Tomiyama, OD, and Kathryn Richdale, OD, PhD
For Review of Optometry, January 2019
Release Date: January 15, 2019
Expiration Date: January 15, 2022
Estimated time to complete activity: 2 hours
Jointly provided by Postgraduate Institute for Medicine and RGVCE
Educational Objectives: After completing this activity, the participant should be better able to:
- Interpret the worldwide increase in myopia prevalence from an epidemiologic perspective.
- Discuss the normal pathophysiologic process of emmetropization and its relation to myopization.
- Explain the likely environmental and behavioral causes for the increase in myopia prevalence.
- Use knowledge of the genetic factors associated with susceptibility for myopia and high myopia to identify and treat patients.
- Recognize how genetic causes relate to environmental factors in regard to light-induced signaling in myopia development.
Target Audience: This activity is intended for optometrists engaged in the care of patients with myopia.
Accreditation Statement: In support of improving patient care, this activity has been planned and implemented by the Postgraduate Institute for Medicine and RGVCE. Postgraduate Institute for Medicine is jointly accredited by the Accreditation Council for Continuing Medical Education, the Accreditation Council for Pharmacy Education, and the American Nurses Credentialing Center, to provide continuing education for the healthcare team. Postgraduate Institute for Medicine is accredited by COPE to provide continuing education to optometrists.
Faculty/Editorial Board: Erin Tomiyama, OD, course instructor, preceptor/teaching fellow, Kathryn Richdale, OD, PhD, associate professor, University of Houston College of Optometry.
Credit Statement: This course is COPE approved for 2 hours of CE credit. Course ID is 60198-PB. Check with your local state licensing board to see if this counts toward your CE requirement for relicensure.
Disclosure Statements:
Dr. Tomiyama: Consulting fees from GPLI Residents Advisory Board.
Dr. Richdale: Contracted research for Alcon and Euclid Planners.
Managers and Editorial Staff: The PIM planners and managers have nothing to disclose. The RGVCE planners, managers and editorial staff have nothing to disclose.
Most parents think genetics are the only reason for their child’s nearsightedness. But more and more children with only one myopic parent, or even no myopic parents, are becoming nearsighted. Clearly, more than genetics is involved in the myopia epidemic.
This article reviews the current understanding of both genetic and environmental associations of myopia, and discusses how to educate parents about why more children are becoming nearsighted, and what can be done about it.
Worldwide Increase
Myopia was previously believed to be a benign error of refractive power. But as more children become nearsighted, myopia is being recognized as a worldwide epidemic with significant health consequences. The rapid increase in prevalence of myopia and consequential risk for pathologic visual conditions, such as cataracts, glaucoma and retinal detachment, make myopia a significant concern for today’s parents.
Higher levels of myopia increase the risk for ocular disease, but even low levels of myopia (-0.75 to -3.00D) are associated with three to four times greater risk of retinal detachment.6 The definition of high myopia varies from study to study, but is usually defined as refractive error of -5.00D or -6.00D or higher.6 Myopia greater than -6.00D increases the risk of retinal detachment by 20 to 80 times that of a non-myope.6
Even though high myopia is the greater threat, we must aim to decrease the amount of myopia for all patients to reduce the overall risk for ocular co-morbidities. Reducing the rate of overall progression by 50% would decrease the prevalence of all levels of myopia, and reduce high myopia by up to 90%.1
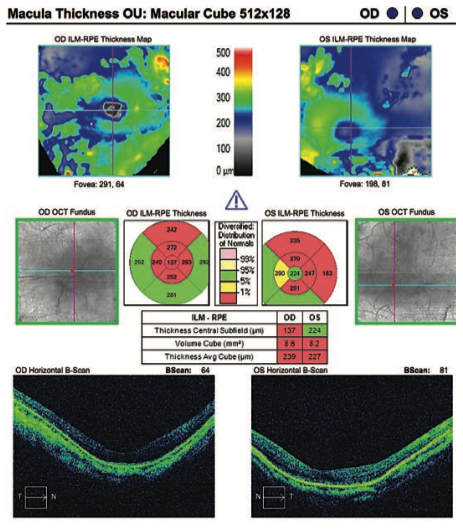
This OCT analysis of a patient with high myopia shows that most of the retinal nerve fiber layer is flagged as abnormal.
Emmetropization and Myopization
The normal pathophysiologic process of emmetropization is such that the eye progresses from hypermetropia in the first years of life until emmetropia is reached in mid-childhood. The goal of emmetropization is for the eye’s corneal and lenticular refractions to match the increasing axial length of the eye during growth. In myopia, the process overshoots emmetropization and results in myopic refractive error.8,9
Research shows myopic children may begin to experience greater axial elongation than emmetropes even three years before the actual onset of myopia.10 In fact, the best determinant of the final refractive error for school-aged children is an age-matched comparison of current refractive state, reported researchers in the Collaborative Longitudinal Evaluation of Ethnicity and Refractive Error (CLEERE) study.11 This was an observational cohort study of ocular development and myopia onset of ethnically diverse children ages six to 13 years. They reported that, if a child is less hyperopic than +0.75D by first grade, the child is at an increased risk to develop myopia.11
We now know that hyperopic defocus (focusing light behind the retina) can stimulate axial elongation. Conversely, myopic defocus (in front of the retina) can slow axial growth.12,13 The peripheral refraction typically varies with the central refraction in that myopes usually have relative hyperopia in the periphery and hyperopes usually have relative myopia in the periphery.14,15
Soft multifocal lenses and orthokeratology lenses are used for myopia control as they shift the peripheral hyperopic defocus to myopic defocus by moving the peripheral focus forward. Corneal topography maps can be used to demonstrate the peripheral plus power that is created with either a soft multifocal lens worn on the eye or after orthokeratology treatment. Multiple studies have now reported good safety and efficacy with these treatment methods.
Environmental and Behavioral Influences
The rapid increase in the prevalence of myopia within the past few decades suggests the involvement of environmental causes, especially when we see such dramatic increases among specific populations and in certain regions. Given this increase in such a short time period, geographic and racial/ethnic differences cannot fully explain the sudden rise in myopia. Ultimately, genetic susceptibility and environmental changes have likely worked in combination to produce the greater prevalence of myopia observed today.1
Research shows that the amount of time a child spends outdoors is directly related to the odds of the child developing myopia.16 While time outdoors is certainly a factor, it’s not certain what aspect of outdoor time directly decreases the risk—could it be the higher light levels, the spectral composition of outdoor light, or the dioptric demand of outdoor viewing?
One study found that children who spent sufficient time outdoors, quantified as more than two hours per day, decreased their risk of myopia even if they also performed a large amount of near work or had two myopic parents.18 The protective effect of time outdoors against myopia was not activity-dependent. Interestingly, this study also reported that there was a greater effect of delaying myopia onset when children performed sports outdoors as compared to doing the exact same sport indoors.18
While there is no magic number, about two hours per day or at least 10 hours per week of outdoor time can have a positive impact for children who haven’t yet developed myopia. Unfortunately, once a child becomes myopic, increasing outdoor time isn’t likely to slow the progression of myopia, according to the limited evidence we have.16
The dioptric mapping (amount of distant vs. near stimuli) of the visual environment could help to explain why more time outdoors may delay the onset of myopia. The dioptric topography of an indoor environment is generally closer and more heterogenous than an outdoor environment, so the eyes are more likely to experience hyperopic defocus indoors.6 Conversely, the outdoor visual environment provides a more uniform field of view on the retina with little dioptric demand.6
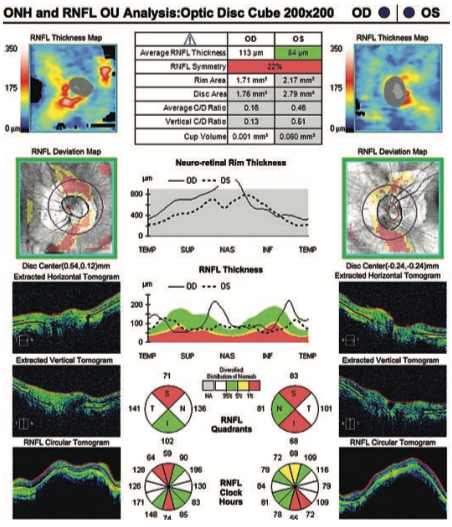
This OCT crosssection reveals a posterior staphyloma—a hallmark sign of pathologic myopia.
Other research suggests that the bright outdoor light triggers a release of dopamine from the retina, inhibiting signals for axial elongation.18 Seasonal variation in the progression of myopia would support this theory, as more axial growth occurs during winter months than during summer months.19
Taken together, studies done in Japan, China and the United States suggest that higher light levels, longer days and higher solar irradiation seem to be associated with slowed myopia progression.19-21
Many challenges to establishing a causal relationship between time outdoors and myopia development and progression exist. For instance, increased time outdoors is indirectly proportional to time indoors, since they are mutually exclusive. Also, children’s activities performed outdoors often involve more physical movement compared with indoors activities, which tend to be more sedentary and involve closer dioptric stimuli. Lastly, many studies examining the amount of time spent outdoors are conduted by survey and are therefore subject to recall bias.16
Researchers have made efforts to use technological advancements to objectively record time spent outdoors and light levels of exposure, but depending on the placement of these devices, they may not truly quantify visual input to the eyes.22 New devices are currently being studied to also record the real-time viewing distances.23
Further studies are needed to gain a better understanding of what factor, or combination of factors, make outdoor time effective in delaying the onset of myopia. For now, we can only present the facts when educating parents and children: increased time outdoors will help delay the onset of myopia, but will not likely have an effect once a child becomes myopic.
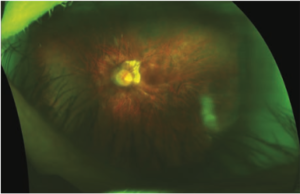
While high levels of myopia have greater risk for pathologic changes, even low levels of myopia (-0.75 to -3.00D) are associated with increased risk for retinal disease.
Research also shows that near work and education level can modify the risk of developing myopia. Myopia progression increases approximately 2% for every diopter of near work.24 The mechanism behind increased near work to increased axial length includes peripheral defocus and biomechanical changes.25 The risk of development and progression of myopia is more correlated with a closer reading distance (less than 20cm) and longer periods of continuous near work (more than 45 minutes) than total duration of near work.26,27
The location in which a child lives can also influence myopia risk. One meta-analysis shows that children in urban environments had 2.6 times increased risk of myopia compared with children in rural areas.33 This finding may be related to the observation that children in urban environments may spend more time indoors with greater near work demands.
Environmental and behavioral factors appear to play important roles in the development and progression of myopia. But if we recommend modifiable behaviors that may slow the onset of myopia, we must also appreciate the important role that genetics play.
Genetic Factors
The risk of a child becoming myopic when one parent is myopic is approximately 1.5 times that for children with no myopic parents. That risk doubles to three times if both parents are myopic. The degree of the parents’ myopia also plays an important factor in the development and final refractive error of the child.34 Parents can readily understand these relative risk factors, but, unfortunately, myopia genetics is not as straightforward as one might expect.
Twin studies. With the exception of rare autosomal dominant conditions such as Marfan syndrome, most “school age” myopia has a complex genetic and environmental etiology.35 Heritability is the proportion of variation in a trait (myopia) that can be attributed to genetic factors. In fact, the first true twin study of heritability was actually conducted by a German ophthalmologist, Walter Jablonski, who identified a greater “within pair” difference in refractive error for fraternal vs. identical twins.36
Some of the more recent twin studies reported myopia heritability ranging from 75% to 95%; however, twin studies are known to overestimate true heritability due to model assumptions and confounding factors such as a largely shared environment.37 Family studies can add more information about the complexity of genetic covariance and environmental factors. Large family studies have reported heritability of refractive error to be 50% to 60%, and a meta-analysis of both twin and family studies suggest a myopia heritability of about 71%.37
Genetic linkage studies. These use familial information to map locations on genes that may be related to a trait. The first myopia-related locus identified was MYP1, which is related to high myopia and located on the X chromosome.38 While other loci have been identified, linkage studies often highlight large regions of the gene, making further analysis challenging.
Candidate gene analyses have identified more than 100 potential myopia-related genes based on known biological function. For example, genes involved in ocular development and growth, as well as scleral remodeling, have been identified as potential candidate genes for myopia.38
Unfortunately, the candidate gene approach is limited by current knowledge of gene function. Both linkage studies and candidate gene studies have reported a possible association between high myopia and the paired box 6 (PAX6) gene.39,40 The PAX6 gene is considered a master gene involved in many facets of normal eye development.
Genome-wide association studies (GWAS). This approach compares the genetic information across thousands (or tens or hundreds of thousands) of individuals to identify small differences potentially related to a specified trait. The strength of this approach is that it can explore the entire genome, and thus doesn’t rely on a prior knowledge of function.
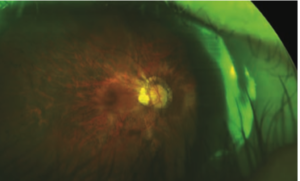
High levels of myopia (-5.00D or -6.00D or greater) increase the risk for ocular disease. Note the peripapillary atrophy and macular changes in this myopic eye
Two of the largest GWAS groups in myopia, the commercial 23andMe and the academic Consortium for Refractive Error and Myopia (CREAM), recently combined efforts to conduct a GWAS meta-analysis of more than 160,000 participants, which identified 161 potential gene loci involved in myopia.41 Some of the loci identified include those involved in dopamine and light processing. These genetic findings provide further support for mechanisms of myopia development involving light exposure, defocus and contrast.
Despite these important findings informing potential genetic locations and mechanisms of myopia development, these studies have identified less than 5% of the variation in refractive error due to genetic variants.42 Clearly, the dramatic rise in myopia cannot be due to genetics alone, but is likely due to a combination of both genetic susceptibility and increased environmental triggers.
Gene-environment interaction studies. To study both genetic susceptibility and increased environmental triggers, gene-environment interaction studies have been used to explore some of the more well-established links with myopia, and have found associations with educational level and near work.42 Mendelian randomization uses genes with known functions as surrogates for environmental factors to explore the cause and effect of environmental exposure on the trait of interest. This is the technique that was used to estimate that every additional year of education was associated with a -0.27D increase in myopia.32
Epigenetics. The study of epigenetics has also been used to explore modifications in gene expression or gene activity due to environmental factors. Of note, one recent epigenetics study found that variants in a micro RNA (miRNA 328) modified PAX6 expression, which may be a potential target for myopia treatment.43 The contribution of genetics directly on the development of myopia may not be comprehensive, but one’s genes may play an important role in one’s susceptibility to environmental factors.44
Discussing ‘Nature and Nurture’ with Parents
When discussing myopia with parents, establish what physicians know about myopia and its progression. Parents should also understand the risks and consequences of developing myopia, specifically the risk for developing retinal holes, tears or detachments, myopic maculopathy, glaucoma and early cataracts.6 While myopia has a genetic component, modifiable behaviors impact the development as well as the rate of progression of myopia. Explaining these modifications can help parents understand that environment does play a role in the development of myopia and actions can be taken to help delay the onset and progression of their child’s nearsightedness.
Online calculators are available to help communicate with patients and parents about the need for initiating treatment. Three primary forms of off-label treatment are available in the United States for myopia control: atropine, soft multifocal contact lenses and orthokeratology. As practitioners, it is our responsibility to educate parents on what myopia is, how it develops, what causes it to progress, and how we may be able to delay its onset and slow its progression. Present all treatment options, as well as their associated risks and benefits.
We may never fully understand the exact causal relationship between our genetics and the many environmental factors related to myopia due to their tight inter-relationship and our inability to restrict natural human development. However, continued genetic and environmental research will likely shed more light on the interaction between nature and nurture, and perhaps bring this epidemic under control.
Dr. Tomiyama recently completed a cornea and contact lens residency at the University of Houston College of Optometry (UHCO) in Houston. Dr. Richdale is an associate professor at UHCO.
- Holden BA, Fricke TR, Wilson DA, et al. Global prevalence of myopia and high myopia and temporal trends from 2000 through 2050. Ophthalmology. 2016;123(5):1036-42.
- Vitale S, Sperduto RD, Ferris FL 3rd. Increased prevalence of myopia in the United States between 1971-1972 and 1999-2004. Arch Ophthalmol. 2009;127(12):1632-9.
- Bourne RR, Stevens GA, White RA, et al; Vision Loss Expert Group. Causes of vision loss worldwide, 1990-2010: a systematic analysis. Lancet Glob Health. 2013;1(6):e339-49.
- Smith TS, Frick KD, Holden BA, et al. Potential lost productivity resulting from the global burden of uncorrected refractive error. Bull World Health Organ. 2009;87(6):431-7.
- Lim MC, Gazzard G, Sim EL, et al. Direct costs of myopia in Singapore. Eye (Lond). 2009;23(5):1086-9.
- Flitcroft DI. The complex interactions of retinal, optical and environmental factors in myopia aetiology. Prog Retin Eye Res. 2012;31(6):622-60.
- Ohno-Matsui K, Yoshida T, Futagami S, et al. Patchy atrophy and lacquer cracks predispose to the development of choroidal neovascularisation in pathological myopia. Br J Ophthalmol. 2003;87(5):570-3.
- Wildsoet CF. Active emmetropization–evidence for its existence and ramifications for clinical practice. Ophthalmic Physiol Opt. 1997;17(4):279-90.
- Morgan I, Rose K. How genetic is school myopia? Prog Retin Eye Res. 2005;24(1):1-38.
- Mutti DO, Hayes JR, Mitchell GL, et al; CLEERE Study Group. Refractive error, axial length, and relative peripheral refractive error before and after the onset of myopia. Invest Ophthalmol Vis Sci. 2007;48(6):2510-9.
- Zadnik K, Sinnott LT, Cotter SA; Collaborative Longitudinal Evaluation of Ethnicity and Refractive Error (CLEERE) Study Group. Prediction of juvenile-onset myopia. JAMA Ophthalmol. 2015;133(6):683-9.
- Sankaridurg P, Holden B, Smith E 3rd, et al. Decrease in rate of myopia progression with a contact lens designed to reduce relative peripheral hyperopia: One-year results. Invest Ophthalmol Vis Sci. 2011;52(13):9362-7.
- Smith EL 3rd, Hung LF, Arumugam B. Visual regulation of refractive development: insights from animal studies. Eye (Lond). 2014;28(2):180-8.
- Millodot M. Effect of ametropia on peripheral refraction. Am J Optom Physiol Opt. 1981;58(9):691-5.
- Mutti DO, Sholtz RI, Friedman NE, Zadnik K. Peripheral refraction and ocular shape in children. Invest Ophthalmol Vis Sci. 2000;41(5):1022-30.
- Xiong S, Sankaridurg P, Naduvilath T, et al. Time spent in outdoor activities in relation to myopia prevention and control: a meta-analysis and systematic review. Acta Ophthalmol. 2017;95(6):551-566.
- Wu PC, Tsai CL, Wu HL, et al. Outdoor activity during class recess reduces myopia onset and progression in school children. Ophthalmology. 2013;120(5):1080-5.
- Rose KA, Morgan IG, Ip J, et al. Outdoor activity reduces the prevalence of myopia in children. Ophthalmology. 2008;115(8):1279-85.
- Gwiazda J, Deng L, Manny R, Norton TT; COMET Study Group. Seasonal variations in the progression of myopia in children enrolled in the correction of myopia evaluation trial. Invest Ophthalmol Vis Sci. 2014;55(2):752-8.
- Fujiwara M, Hasebe S, Nakanishi R, et al. Seasonal variation in myopia progression and axial elongation: an evaluation of Japanese children participating in a myopia control trial. Jpn J Ophthalmol. 2012;56(4):401-6.
- Donovan L, Sankaridurg P, Ho A, et al. Myopia progression in Chinese children is slower in summer than in winter. Optom Vis Sci. 2012;89(8):1196-202.
- Dharani R, Lee CF, Theng ZX, et al. Comparison of measurements of time outdoors and light levels as risk factors for myopia in young Singapore children. Eye (Lond). 2012;26(7):911-8.
- Ostrin LA, Sajjadi A, Benoit JS. Objectively measured light exposure during school and summer in children. Optom Vis Sci. 2018;95(4):332-342.
- Huang HM, Chang DS, Wu PC. The association between near work activities and myopia in children—A systematic review and meta-analysis. PLoS One. 2015;10(10):e0140419.
- Ghosh A, Collins MJ, Read SA, Davis BA, Chatterjee P. Axial elongation associated with biomechanical factors during near work. Optom Vis Sci. 2014;91(3):322-9.
- Li SM, Li SY, Kang MT, et al; Anyang Childhood Eye Study Group. Near work related parameters and myopia in Chinese children: The Anyang Childhood Eye Study. PLoS One. 2015;10(8):e0134514.
- Ip JM, Saw SM, Rose KA, et al. Role of near work in myopia: Findings in a sample of Australian school children. Invest Ophthalmol Vis Sci. 2008;49(7):2903-10.
- Dolgin E. The myopia boom. Nature. 2015;519(7543):276-8.
- Rose KA, French AN, Morgan IG. Environmental factors and myopia: Paradoxes and prospects for prevention. Asia Pac J Ophthalmol (Phila). 2016;5(6):403-410.
- Ben-Simon GJ, Peiss M, Anis E, et al. Spectacle use and reduced unaided vision in third grade students: a comparative study in different educational settings. Clin Exp Optom. 2004;87(3):175-9.
- Kinge B, Midelfart A, Jacobsen G, Rystad J. The influence of near-work on development of myopia among university students. A three-year longitudinal study among engineering students in Norway. Acta Ophthalmol Scand. 2000;78(1):26-9.
- Mountjoy E, Davies NM, Plotnikov D, et al. Education and myopia: assessing the direction of causality by mendelian randomisation. BMJ. 2018;361:k2022.
- Rudnicka AR, Kapetanakis VV, Wathern AK, et al. Global variations and time trends in the prevalence of childhood myopia, a systematic review and quantitative meta-analysis: Implications for aetiology and early prevention. Br J Ophthalmol. 2016;100(7):882-890.
- Zhang X, Qu X, Zhou X. Association between parental myopia and the risk of myopia in a child. Exp Ther Med. 2015;9(6):2420-2428.
- Aubart M, Gazal S, Arnaud P, et al. Association of modifiers and other genetic factors explain Marfan syndrome clinical variability. Eur J Hum Genet. 2018;26(12):1759-1772.
- Liew SH, Elsner H, Spector TD, Hammond CJ. The first “classical” twin study? Analysis of refractive error using monozygotic and dizygotic twins published in 1922. Twin Res Hum Genet. 2005;8(3):198-200.
- Sanfilippo PG, Hewitt AW, Hammond CJ, Mackey DA. The heritability of ocular traits. Surv Ophthalmol. 2010;55(6):561-83.
- Jacobi FK, Pusch CM. A decade in search of myopia genes. Front Biosci (Landmark Ed). 2010;15:359-72.
- Hammond CJ, Andrew T, Mak YT, Spector TD. A susceptibility locus for myopia in the normal population is linked to the PAX6 gene region on chromosome 11: a genomewide scan of dizygotic twins. Am J Hum Genet. 2004;75(2):294-304.
- Tang SM, Rong SS, Young AL, et al. PAX6 gene associated with high myopia: a meta-analysis. Optom Vis Sci. 2014;91(4):419-29.
- Tedja MS, Wojciechowski R, Hysi PG, et al. Genome-wide association meta-analysis highlights light-induced signaling as a driver for refractive error. Nat Genet. 2018;50(6):834-848.
- Verhoeven VJ, Hysi PG, Wojciechowski R, et al. Genome-wide meta-analyses of multiancestry cohorts identify multiple new susceptibility loci for refractive error and myopia. Nat Genet. 2013;45(3):314-8.
- Chen KC, Hsi E, Hu CY, et al. MicroRNA-328 may influence myopia development by mediating the PAX6 gene. Invest Ophthalmol Vis Sci. 2012;53(6):2732-9.
- Lim LT, Gong Y, Ah-Kee EY, et al. Impact of parental history of myopia on the development of myopia in mainland china school-aged children. Ophthalmol Eye Dis. 2014;6:31-5.













