October 2, 2019
By Dwight Akerman, OD, MBA, FAAO
Chief Medical Editor, Review of Myopia Management
 Atropine is a non-selective anti-muscarinic agent that has a high affinity to M1-M5 receptors in the pupillary sphincter and ciliary muscle resulting in mydriasis and cycloplegia. Numerous clinical studies, primarily conducted on East Asian children, have explored the effect of topical low-dose atropine on myopia progression. However, its use in children of European ancestry remains controversial, and substantial evidence is sparse. The Oxford Centre for Evidence Based Medicine (OCEBM) classifies the evidence for atropine therapy as level I for East Asian populations but only as level IV in non-Asian populations.
Atropine is a non-selective anti-muscarinic agent that has a high affinity to M1-M5 receptors in the pupillary sphincter and ciliary muscle resulting in mydriasis and cycloplegia. Numerous clinical studies, primarily conducted on East Asian children, have explored the effect of topical low-dose atropine on myopia progression. However, its use in children of European ancestry remains controversial, and substantial evidence is sparse. The Oxford Centre for Evidence Based Medicine (OCEBM) classifies the evidence for atropine therapy as level I for East Asian populations but only as level IV in non-Asian populations.
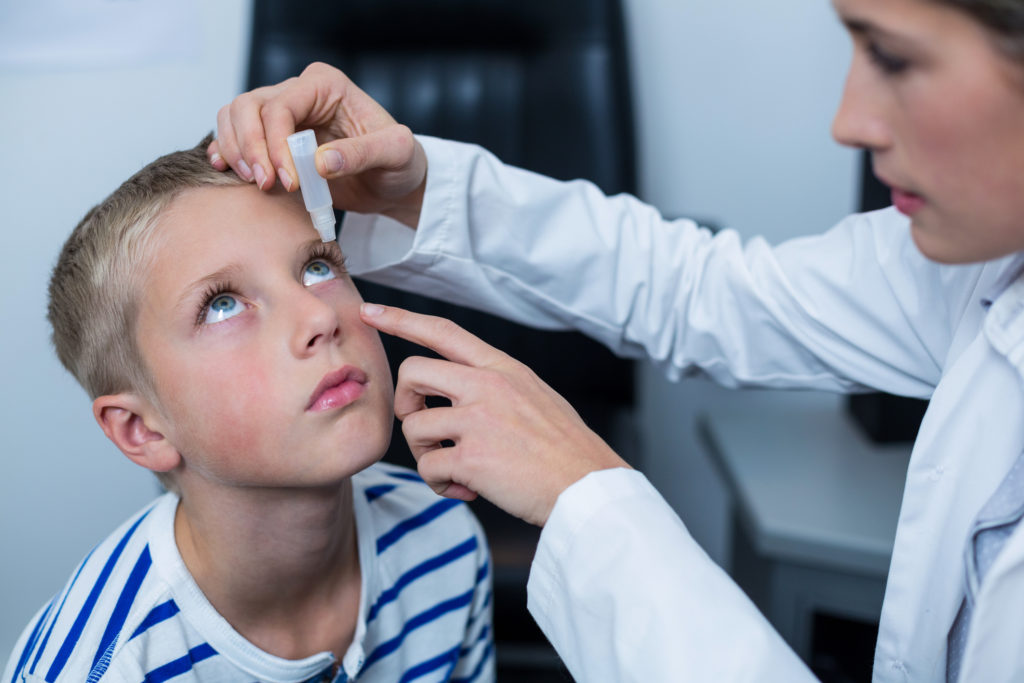
In this study conducted in Germany, 56 children, who had a median age of 11 years (range 6 to 17 years), were analyzed after 12 months of topical treatment with 0.01% preservative-free atropine in both eyes at bedtime every day. Efficacy was assessed during treatment every six months. In a subset of 20 patients, treatment of the second eye was delayed by one day to enable a controlled safety assessment of side effects such as pupil dilation, hypo-accommodation, and near vision reduction.
Prior to treatment, the mean myopia progression was estimated at 1.05D/year; after 12 months of treatment with 0.01% atropine, it was 0.40D/year (p < 0.0001). This represents a 62% reduction in the progression of refractive myopia. The only consistently measurable side effect was the induction of 1mm pupil dilatation, which was noticeable only in comparison to the non-treated eye during the safety investigation.
The researchers concluded that topical low-dose atropine appears to be safe and effective in a cohort of European schoolchildren. These data should motivate researchers to conduct more randomized clinical trials, especially on non-Asian children.
ABSTRACT
A Pilot Study on the Efficacy and Safety of 0.01% Atropine in German Schoolchildren with Progressive Myopia
Joachimsen L, Böhringer D, Gross NJ, Reich M, Stifter J, Reinhard T, Lagrèze WA.
Introduction: Although the interest is growing in topical low-dose atropine to control myopia in schoolchildren worldwide, its use in children of European ancestry remains controversial and solid evidence is sparse. The Oxford Centre for Evidence Based Medicine (OCEBM) classifies the evidence for this therapy as level I for East Asian populations, but only level IV in non-Asian populations.
Methods: Fifty-six children, aged a median of 11 years (range 6-17), were analysed after 12 months of topical treatment with 0.01% preservative-free atropine in both eyes at bedtime every day. Efficacy was assessed during treatment every 6 months. In a subset of 20 patients, treatment of the second eye was delayed by 1 day to enable a controlled safety assessment of side effects such as pupil dilation, hypo-accommodation, and near vision reduction.
Results: Prior to treatment, the mean myopic progression was estimated as 1.05 D/year; after 12 months of treatment with 0.01% atropine, it was 0.40 D/year (p < 0.0001). The only consistently measurable side effect was the induction of 1 mm pupil dilatation, which was only noticeable in comparison to the non-treated eye during the safety investigation.
Conclusions: Topical low-dose atropine appears to be safe and efficacious also in a cohort of European schoolchildren. These data should motivate researchers to conduct more randomised clinical trials
Source: Joachimsen, L., Böhringer, D., Gross, N. J., Reich, M., Stifter, J., Reinhard, T., & Lagrèze, W. A. (2019). A Pilot Study on the Efficacy and Safety of 0.01% Atropine in German Schoolchildren with Progressive Myopia. Ophthalmology and therapy, 1-7.
DOI: 10.1007/s40123-019-0194-6